July 1, 2013
Watch the video bove to see examples of how software-driven simulation has made the leap from the automotive and aerospace industries and into the biomedical field.
Martin Larsen, a simulation specialist at Oticon, uses ANSYS Mechanical, the same software automakers would use to reduce brake squeal or test noise, vibration and harshness (NVH). But Larsen doesn’t design vehicles; he designs hearing aids.
Wind Feng, an R&D engineer at MicroPort, uses Dassault Systemes SIMULIA. Aerospace manufacturers use the same program to optimize hypersonic re-entry vehicles. Feng, however, happens to work in orthopedics.
Darryl D’Lima, M.D., Ph.D., from Scripps Research Institute, has more than 10 years of experience in MSC Software’s Marc, a program widely used in transportation and energy. But D’Lima isn’t researching transmissions and gears; he studies cartilage injury, stem cells, and joint wear.
These are just a few examples of the biomedical profession’s flirtation with simulation technologies.
Manufacturing activities in automotive and aerospace fuel R&D and commerce in the simulation sector. Industry titans like GM, Ford, Boeing and Airbus play a big part in shaping the technology and the workflow. Because the software programs have gone through cycles of trial and error over the years, the latest generation of solvers can replicate car- and plane-related phenomena with reliable fidelity. Computational fluid dynamics (CFD), structural dynamics, preprocessing, post-processing and meshing are part of automotive and aerospace engineers’ daily vocabulary.
For doctors, physicians, and researchers in life sciences, however, simulation vocabulary might as well be Greek or Latin. Come to think of it, Greek or Latin might be easier for doctors to grasp, because it’s the root of many medical terms.
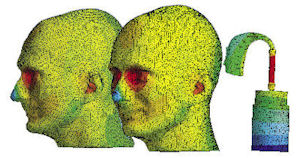
Oticon, a hearing aid manufacturer, uses ANSYS to simulate how its products fit and function inside ear canals. |
Can these new users easily adopt programs initially designed with another industry in mind? Can the programs used to simulate heat distribution and mechanical operations inside an engine block be used to simulate hearing aids and knee joints? Is specifying the right viscosity, thermal conductivity and tensile strength for human tissue and blood as simple as picking them out of a dropdown menu in a library and adjusting their properties? The answers to these questions suggest simulation software makers have a lot to learn from the new professions they’re courting.
Group Treatment Not Available
With a strong presence in healthcare as well as product lifecycle management (PLM), Siemens makes not only medical devices, but also the software used to simulate them. The symbiosis between its device and simulation software divisions is reflected in the ease with which you can reuse magnetic resonance imaging (MRI) data inside NX, the company’s leading computer-aided design/manufacturing/engineering software suite. That’s according to Martin Kuessner, global lead of NX CAE business development at Siemens PLM Software.
“In medical device, pharmaceutical and biomedical, there’s a lot of simulation going on,” Kuessner says. “It’s rapidly increasing. However, [the use] is not as straightforward as in other industries. It’s much more complex, much more diverse. Therefore, it might not have the visibility and the penetration it deserves.”
Automotive components have become highly standardized; consequently, developing a custom automotive simulation package is relatively easy. Not so with medical devices, says Kuessner.
“A stent, a heart valve, a tablet dispenser, a liquid container—they have different challenges, demand different degrees of accuracy,” he points out. Neither Siemens nor any other simulation software maker can possibly come up with a single “prefabricated, preconfigured simulation solution” to accommodate this of diversity. However, each application area can be covered by its own customized environment for easier access to simulation, he says.
Customization: Pros and Cons
Oticon’s Larsen simulates the way sound waves and vibrations travel through the ear canal using ANSYS Mechanical. Larsen and his colleagues tweaked the software to fit their own workflow, based on their accumulated understanding of the inner working of ear canals. They load the software with custom material databases. These, he says, are closely guarded secrets among the handful of companies competing for dominance in hearing aid design.
In its deployment of ANSYS Mechanical, Oticon pays particular attention to acoustic loss, resulting from the use of tiny tubes in hearing aids. Larsen and his colleagues thought about developing a general user interface for acoustic simulation, but found out they didn’t need to—ANSYS beat them to the punch by introducing the feature in Version 13. By Version 14.5, ANSYS has come up with acoustic loss analysis. Larsen hopes to see more developments to this function in the future.
In some instances, the requirements of the medical device maker may be too specialized for the general-purpose simulation package to address. In that case, the user has no choice but to develop custom codes or plug-ins. “We try to do everything in ANSYS if we can,” says Larsen. “We want to spend our time developing hearing aids, not custom software. That said, we have developed own tools as well.”
In the past few years, Chinese medical device and implant manufacturer MicroPort has closely collaborated with DS SIMULIA in structure optimization, kinematics simulation and fatigue analysis. MicroPort’s Feng says, “Every customer has its own requirement for simulation. We’ve made a customized process for a cardiovascular implant, and find it efficient and useful.”
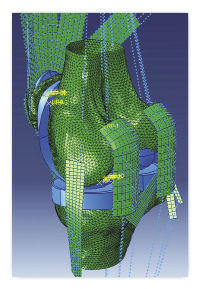
This meshed FEA model of a Total Knee Replacement (TKR) is ready for analysis in the Abaqus Knee Simulator AKS from Dassault Systemes SIMULIA. The 2D rectangular strips and the vertical lines are tendons. The TKR components are in blue. The two large green knobs are the tibia and the femur. |
At least when it comes to knee implants, MicroPort won’t need to write custom code. DS SIMULIA came up with the Abaqus Knee Simulator (AKS), based on its general-purpose finite element analysis (FEA) program Abaqus. The company describes the module as “a validated computational modeling tool for performing basic to advanced knee implant analyses and simulations. This tool offers five fast and easy-to-setup workflows which reduce your reliance on time-consuming trials and expensive lab equipment, while still meeting regulatory requirements.”
Feng says simulation helps MicroPort “reduce physical prototyping and shorten mechanical and fatigue testing times. More importantly, it helps us evaluate the biomechanics performance of our design without conducting expensive and lengthy physical tests.”
MicroPort uses DS SIMULIA’s AKS to simulate two types of knee implants: fixed bearing and mobile bearing.
“Because of their complexity and loading conditions, setting up these two models could easily have taken a week by hand,” Feng says. “Instead, it took a day or two. The AKS makes it practical for product development companies to perform simulations as thorough as those at research institutes.”
The Language Barrier
Thierry Marchal, ANSYS’ industry director for healthcare, says, “Many of the people in medical device have an engineering background. Often, they came from other industries. Some have worked at places like Boeing, so they know simulation, even know the software.” But he points out a new class of simulation users are emerging: clinical technicians, surgeons and physicians. “These people are so busy operating on patients and saving lives, there’s no way they can invest the time to learn the software,” says Marchal. For them, the language in simulation could be a significant barrier. That language consists of material tensile strength, outside pressure, internal temperature, fluid turbulence model, and other computable values.
Siemens’ Kuessner sees value in a simulation program with customized input fields. “That way, you won’t be talking about Young’s modulus, but instead, you can ask for bone types [as input],” he reasons. “At the end of the day, you’re still dealing with engineering calculation—strain energy, stress and so on—but the interface should be customized for the user.”
Such a concept might be difficult to implement at the software level, however. Ashley Peterson, principal R&D engineer at Medtronic, a medical device manufacturer, points out that there’s currently no good way to translate patient data into computable simulation values. Even if it were possible, he wonders whether it would be wise.
“If you take, say, a patient’s age and correlate it to a certain blood flow or viscosity, you’re saying, that’s generally what you find. But that’s not true in the cases where the patient has a certain disease, or if they have a slightly elevated blood pressure,” he points out. “If you use averages, and remove too many of the details, you may be reducing the accuracy of the simulation.”
Carlos Olguin, head of the bio-nano programmable matter group at Autodesk, says, “In my view, it is less about replacing and more about mapping and correlating standard medical terms and measurements to the engineering vocabulary. And the engineering vocabulary itself will also change, depending on the metric scale at which a simulation is performed.”
Olguin offers as an example how at the molecular scale, gravity plays a less important role, while diffusion is emphasized: “As one goes even further down in scale, then Newtonian physics are gradually replaced by quantum physics.”
When Metal Meets Flesh
Peterson and his colleagues at Medtronic employ CD-adapco’s STAR CCM+ (for CFD) to develop and test medical devices. In some simulation scenarios, he needs to understand not just how the medical device operates, but how it interacts with organic matter. To simulate an endovascular stent graft, for example, requires simulating the way the device fits inside a patient’s aortic aneurysm and interacts with the blood and vasculature.
In another division of Medtronic, the company uses ANSYS software to simulate how an implanted device behaves when a patient is subjected to an MRI scan. Mariya Lazebnik, Ph.D., senior scientist at Medtronic, points out, “Right now, patients with implantable devices cannot have an MRI scan because of the harmful interaction between the device and the scanner.”
During an MRI scan, body temperature rises, which affects the implanted device. “This is a complex problem,” says Lazebnik. “The size and shape of the patient will influence the magnitude of that interaction.” Medtronic uses coupled physics—electromagnetic and thermal—in ANSYS to simulate these scenarios.
Using MSC Software’s Marc nonlinear FEA software, researchers at Scripps simulated wear-related complications in orthopedic joint replacements, in scenarios where metals, polymers and biological materials literally interact with one another. Surprisingly, D’Lima reveals he and his colleagues are using Marc as-is, with little or no customization. He is considering using MSC Adams for biomechanics as a next step.
“We’ve solved the geometry problem with better imaging tools,” says D’Lima. “We can now construct accurate surfaces from CT scans, and then turn them into meshes that the simulation solvers can handle. Getting valid forces and motions are still a challenge. We have machines that measure friction and wear, but material property is a bigger challenge. We can test the heck out of metals and artificial polymers used for implants, but getting the properties of bones, tendons, ligaments ... that’s not easy.”
It’s a bit easier for Oticon, Larsen reports, because hearing aids are inserted into the ear canal; they don’t get embedded below the skin or muscles. Larsen doesn’t need to worry about simulating tissues. Rather, his challenge lies in customization.
“Each ear canal is very individual,” he explains. “No two are alike. We have some very good information on certain types of ear canals. Then there are others we collect using special methods. The problem when obtaining ear canal geometries is to get close to the eardrum, since you can seriously hurt the person if you touch their eardrum.”
Ongoing industry efforts include scanning different ear canals and studying them to understand sound-distribution directions, Larsen notes. That fuels his use of ANSYS Mechanical.
“We cooperate with the Technical University of Denmark (DTU), where they can generate 3D models of heads from photos,” he adds. The head with the hearing aid shown on page 16 (based on a scan of his own head, Larsen reveals) has been scanned with this method.
Biomaterial Properties
Perhaps the biggest hurdle in simulating biomedical events is the lack of material data. In most cases, the simulation software won’t give you the option to pick a suitable type of bone, tissue or blood from a dropdown menu. (It would if you were simulating a plastic, steel or metal part.) Biomaterial data doesn’t exist in a form that can be readily delivered inside current software. The simulation experts and software developers interviewed for this article don’t always agree on custom biomedical modules’ benefits, or on how to make software interfaces more approachable—but they all lament the lack of biomaterial data.
Siemens’ Kuessner describes the way it currently works: “The material laws [such as the fundamentals of Young’s modulus] are part of the simulation code, the FEA or CFD program. We provide very sophisticated material laws,” he points out. “But it’ll be the responsibility of the user to put [the sample material] into a test machine if possible, or measure it in situ, calibrate the material, and bring life to the mathematics.” This, he concedes, could be a big bottleneck for users.
ANSYS’ Marchal says biomedical problems are more complex.
“Look at cardiovascular modeling, or simulating blood flow in deformed arteries. When you’re talking about blood, its properties are not the same as air for airplanes or water for turbines that can be modeled with a constant viscosity and density,” Marchal continues. “Blood properties are not constant; they change with [the patient’s] activities, pathologies and conditions such as age, sex and health. Muscles and soft-tissues are not like metal, and need more than a constant Young modulus and Poisson coefficient to be properly modeled. It is therefore crucial to collaborate with clinicians to start building database of patient specific material properties.
“It’s easy to develop [a specialized module], but if used inappropriately, it can give you the wrong answer while you would trust it because this is the result of simulation,” he says.
Where Do We Go from Here?
“Every simulation is wrong. Some are useful,” Siemens’ Kuessner says. It’s his way of reminding us that, at its core, simulation is a simplified representation of a complex event. Nevertheless, the simplified study of nature, he insists, can be a great help in product design.
“Some people say, ‘Oh, we don’t know enough about the materials, so we can’t simulate,’” he says. “I say that’s wrong. You can still do an envelope simulation: Take the worst case, take the best case, and study the range to see where problems can potentially occur.”
In April, the American Journal of Neuroradiology published a paper titled “3D Cine Phase-Contrast MRI at 3T in Intracranial Aneurysms Compared with Patient-Specific Computational Fluid Dynamics.” (Obviously, the language barrier gets worse when biomedical and simulation terms merge.) In the abstract, the authors explain, “CFD has been proved valuable for simulating blood flow in intracranial aneurysms, which may add to better rupture risk assessment. However, CFD has drawbacks such as the sensitivity to assumptions needed for the model The purpose of this study was to compare flow patterns on the basis of 3D PC-MR imaging with CFD estimates.”
It’s just one example of the validation needed to ensure the simulated pixels match what really happens inside your body.
Kenneth Wong is Desktop Engineering’s resident blogger and senior editor. Email him at [email protected] or share your thoughts on this article at deskeng.com/facebook.
Subscribe to our FREE magazine, FREE email newsletters or both!
About the Author
Kenneth Wong is Digital Engineering’s resident blogger and senior editor. Email him at [email protected] or share your thoughts on this article at digitaleng.news/facebook.
Follow DE





