Rapid Tech Firms Aid Medical and Dental Fields
Daily production of tens of thousands of parts takes these applications far beyond the prototyping realm.
Latest News
May 1, 2010
By Pamela J. Waterman
 A custom plastic dental drilling guide made on a 3D Systems SLA machine. This aids dentists in drilling holes for inserting tooth implants. Image courtesy 3D Systems |
In the past year, if you or someone you know has had a dental crown installation, new hearing aid fitted, or electrotherapy treatment, the procedure likely involved an additive manufacturing (AM) process. Jay Leno may get the spotlight for building cool one-off car parts, but the truly eye-opening news in rapid technology (RT) comes from the sheer numbers and variety of medically related pieces now in daily production.
Companies such as 3D Systems, Arcam, EOS, Envisiontec, ExOne, Objet Geometries, Solidscape, Stratasys, and Z Corporation are producing plastic and metal products that are either fully qualified as a tool in a medical or dental procedure, or become a final part for the application itself. DE looks at the latest of these accomplishments, which show no signs of slowing down.
Tools of the Trade, RT Style
With necessity, or possibly frustration, being the mother of invention, it’s no surprise that many medical devices first see light of day as a design by an independent doctor. Taking that rough concept from napkin drawing to initial production generally involves working with a product development company that creates a first article for design and material evaluation. If the prototype seems acceptable, production tooling is ordered, easily starting at $50,000. But what happens three months later when field-testing uncovers design flaws in 2,000 injection-molded parts?
New Terminology Standards for AM Finally a group of RT specialists combining years of hands-on experience have compiled the definitive vocabulary list for the additive manufacturing industry. Now newbies, seasoned engineers, and even trade-magazine editors can work from the same page when discussing such terms as stereolithography, subtractive manufacturing, rapid tooling, or reverse engineering. Score one for SME’s Committee F42 on Additive Manufacturing Technologies. (Report available by mail or download for $33 at astm.org)—PJW |
Stratasys is helping minimize the risk of both time and money by offering a step-by-step AM-based path for product development. The company’s Fortus 3D production systems can build parts from a wide range of production-grade thermoplastic materials, including polycarbonate (PC)-ISO and ABS-M30I versions for direct medical applications.
PC-ISO met the material needs for Ablation Frontiers (now part of Medtronic) to provide early adopters with a real, autoclave-safe defibrillator tool for use in open-heart surgery. Working with Stratasys’ RedEye service bureau in a parallel effort of user evaluation and design refinement, the medical group was able to incorporate seven design changes during beta testing of 10 to 20 parts at a time. The tool has now gone into clinical trials.
The ability to combine an AM strategy with an ISO-10993 certified material offers a crucial advantage. Stratasys product manager Tim Thelin says, “When a product development company does a material search, that’s when the door opens (to using our systems).” A second product in development with the help of RedEye is a suture device made from high strength ABS-M30I. This biocompatible material can be sterilized with gamma- or ethylene-oxide processes, necessary in cases where high-temperature steam is not permitted (e.g., instruments with wiring). The device customer is currently shipping surgically qualified products, generating interest and income that will support larger-scale injection-molded manufacturing.
 Working beta-test version of surgical ablation tool; housing produced on Stratasys Fortus system. Image courtesy Stratasys |
3D Systems’ large family of AM products includes high-end metal-based selective laser sintering (SLS) and plastic-based stereolithography apparatus (SLA) systems. Historically, the company has seen many of its SLA pieces used as surgical planning models. Now, an emerging market for both technologies is that of single-use surgical guides, akin to those used as dental drilling guides, in applications such as orthopedic surgery.
“Our 8000 IPro series has taken accuracy to a whole new level,” says Kevin McAlea, 3D Systems’ general manager and vice president for large-frame systems. “Customization of (these) products lends itself to a high throughput machine. We’re talking about hundreds of thousands per year.” Along with the accuracy, having materials that can be sterilized and meet U.S.P Class 6 tests for various aspects of toxicity has been key to customer interest.
Other medical application areas under study on 3D Systems SLS equipment include metal bone-replacement materials built as porous parts and bone-scaffolding pieces built from high-temperature plastics.
Metal bone-replacements, such as hip and spine implants, have long been a specialty of Arcam, developers of electron beam melting (EBM) systems. EBM parts made of titanium or cobalt-chrome are well-suited to osseointegration, since they can be optimized in terms of pore geometry, pore size, relative density, and roughness. Using Arcam equipment, a growing number of medical technology companies have developed standard and patient-specific products that are CE-certified for surgical implant.
Reducing Timelines
Companies that work with Objet Geometries are completely changing the way they do business because of the speed, accuracy, and smooth finish derived from Polyjet Polymer Jetting technology. One medical customer, Ivivi Health Sciences, develops noninvasive electrotherapy devices, some of which stimulate a patient’s antiinflammatory responses, reducing pain and swelling; other products treat cardiac, neurological, and orthopedic conditions.
 Sample biocompatible scaffold structures built on an Envisiontec 3D-Bioplotter. Image courtesy Envisiontec |
Andre A. DiMino, Ivivi vice president of engineering, manufacturing, and regulatory, says that Objet’s Eden350 system has given Ivivi the ability to create working medical devices overnight that are ready for clinical trials. “We were relying on outside resources and, often, it was taking months to create just one very expensive and extremely delicate prototype,” he explains. using an Objet system cuts an average of five to six weeks off the clinical trial device production process.
 Skull model for surgical preplanning, produced in bright white plastic on ZCorp 3D printer. Image courtesy ZCorp |
Another Objet customer, Orchid Design, a division of Orchid Orthopedic Solutions, works with medical professionals to design, prototype, and test new orthopedic solutions. Since development time is critical during the FDA approval process, Orchid was pleased to find that in-house use of Objet’s Alaris30 Desktop 3D printer let it build prototypes 10 to 20 times faster than before; timelines are now reduced by 20 percent.
Growing Business
The introduction of Z Corporation’s water-curable zp150 plastic material has given a boost to device developers as well as doctors planning complex surgeries. As the company’s strongest, whitest, highest resolution material yet for any of its 3D printing systems, the material produces surgical models that look more like bone when compared to parts made from the earlier zp131 powder.
Z Corporation Vice President of Business Development Scott Harmon notes the benefits of using this material and process: Surgical customers can visually differentiate various tissues involved and “wrap” bone-density maps on the model’s surface, while device developers appreciate the brilliant colors that come from working on a bright-white substrate.
Business with medical service suppliers is growing, with adoption overseas even faster than in the U.S. due to insurance reimbursement issues for pre-surgery planning.
Dental applications form a major share of business for Envisiontec, whose AM systems are primarily based on Texas Instruments’ DLP technology. Operating with a choice of materials suitable for the dental and hearing-aid industries, several thousand Envisiontec systems are in use worldwide. The company’s Digital Dental Printer (DDP) creates investment castings for crowns with WIC300, a material that literally melts out (leaving no ash) and also produces dental drill guides with its biocompatible e-Shell 300 material. For crown wax-ups made on a DDP, the average part cost is $0.20.
Is the U.S. on a par with Europe for medical implant adoption? Additive manufacturingindustry consultant Terry Wohlers, doing research for his annual Wohlers Report on the state of the AM industry, points out that the U.S. acceptance of AM technology for medical applications lags that of Europe. He notes, “ The part that’s most frustrating is the lack of adoption of metal orthopedicimplants . I’m unaware of a single company here in the U.S. that is using AM to manufacture metal implants. A minimum of nine European companies are using AM for this application and six are in production at some level. Europe has granted CE certification for the production of hip implant products, but here in the U.S., the FDA has not yet done so.” Wohlers explains that surgeons are using custom implants from AM on a one-off basis. “If you want to introduce amainstream product, the FDA must get involved, and that hasn’t happened in the U.S.” By contrast, one of Arcam’s European customers has CE certification for an implant product and uses six EBM machines to manufacture more than 25,000 products peryear at one location. “There’s nothing remotely close to this here in the U.S.,” Wohlers observes.—PJW |
Envisiontec also plays a role in the world of computer-aided tissue engineering. Its 3D-Bioplotter fabricates scaffolds from a truly impressive range of materials, from tricalcium phosphate for bone regeneration to the FDA-approved copolymer polylactic-co-glycolic acid (PLGA) as a drug-release mechanism, and from agar to chitosan for soft tissue printing.
Metals and Plastics go Bionic
It’s difficult to name an application area where EOS systems don’t play a prominent role, particularly in medicine. This company continues to expand the material offerings for both its plastic and metal precision laser sintering machines, and so the variety of applications grows too. Specialty instrumentation, knee replacements, and dental implants are just three of the many products now created in ongoing product lines.
EOS’ vision has always been to use its technology to tailor patient-specific products; two relatively new applications are moving that plan forward. Andy Snow, EOS regional sales manager for North America, says, “Some of our customers are utilizing our plastic technology for patient-specific hip and knee cutting guides, in preparation for surgery, with the use of our PA2200 nylon (Class 6 biocompatible) material.”
He adds that another EOS plastic—high-temperature PEEK HP3, originally developed for aerospace applications—has attracted interest in the medical arena. Its outstanding tensile strength (up to 94MPa) makes it a candidate to replace stainless steel and titanium in spinal implants. Titanium use continues to gain in the dental world, particularly for dental implant screws, and cobalt-chrome alloys make an excellent base for porcelain restorations.
Solidscape, a pioneer in fine-detail wax-up parts for casting, continues to market its preXacto line of inkjet droplet systems to the dental industry. Its proprietary Smooth Curvature Printing technology contributed to its being recognized as one of the top ten products of 2009 by Dental Labs Products magazine.
More sources are emerging for AM cost-savings in the dental field. 3D Systems has expanded its product line and material offerings to compete strongly in this arena with its dedicated Top to Bottom Dental business. Their crown jewel (no pun intended) is the role their SLA machines play in creating the molds for Invisalign removable orthodontic appliances. Check out the impressive video (search: Invisalign Manufacturing Juarez Mexico) that shows the 35 SLA machines manufacturing 40,000 parts a day (8 to 10 million pieces per year) on which to thermoform the final customized sleeves.
Hearing aid manufacturing (in thousands per day) forms the next largest market, but interest from the general dentistry segment is increasing. Lee Dockstader, vice president of 3D Systems dental segment, explains that many factors are coming together to make this an excellent time to use AM. “The manual way of taking impressions has many variables; intra-oral scanners remove the variables. There’s new software, there’s impression scanners, and the big players use stereolithography to produce the models (for building a crown, etc.) When they take a crown off the model, it fits every time.”
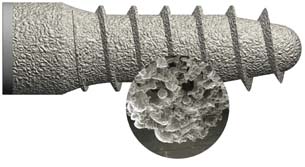 Porous titanium dental implant screw, with excellent osteointegration characteristics, created on an EOS M270 system. Image courtesy EOS |
This level of automation allows dental labs to keep their work from going overseas, saving the mailing time previously required, and the dentist time in fitting the crown to the patient. Other 3D Systems options include its selective laser melting systems for metal copings and implants, as well as the new ProJet MP3000 for $65K.
For more than ten years ExOne and its various divisions, from ProMetal to Imagen, have been involved in printing precious metals, particularly for dental applications. While Imagen is currently undergoing a business reorganization, ProMetal engineers have been busy supporting ADA Technologies’ prosthetics R&D division, PhysioNetics. They make the basic pieces of the V2P Variable Pinch Force prehensor, a cable-controlled arm prosthetic that allows a wide range of grip strengths. Check out the video demo.
AM: Medical Business as Usual?
Just as the dental and hearing-aid worlds now use AM as a regular way of doing business, more medical apps are evolving. Product development companies continue to make the connection between a great new idea and this great way to build it, and more manufacturing companies are treating it as a standard mode of operation. As 3D Systems’ McAlea says, “Seeing some of these applications emerge is truly remarkable—what we can get from an accuracy point of view, I didn’t really think was possible five years ago. It’s really gratifying.”
More Info:
3D Systems
Contributing Editor Pamela J. Waterman, DE’s simulation expert, is an electrical engineer and freelance technical writer based in Arizona. You can send her e-mail to [email protected].
Subscribe to our FREE magazine, FREE email newsletters or both!
Latest News
About the Author
Pamela Waterman worked as Digital Engineering’s contributing editor for two decades. Contact her via .(JavaScript must be enabled to view this email address).
Follow DE





